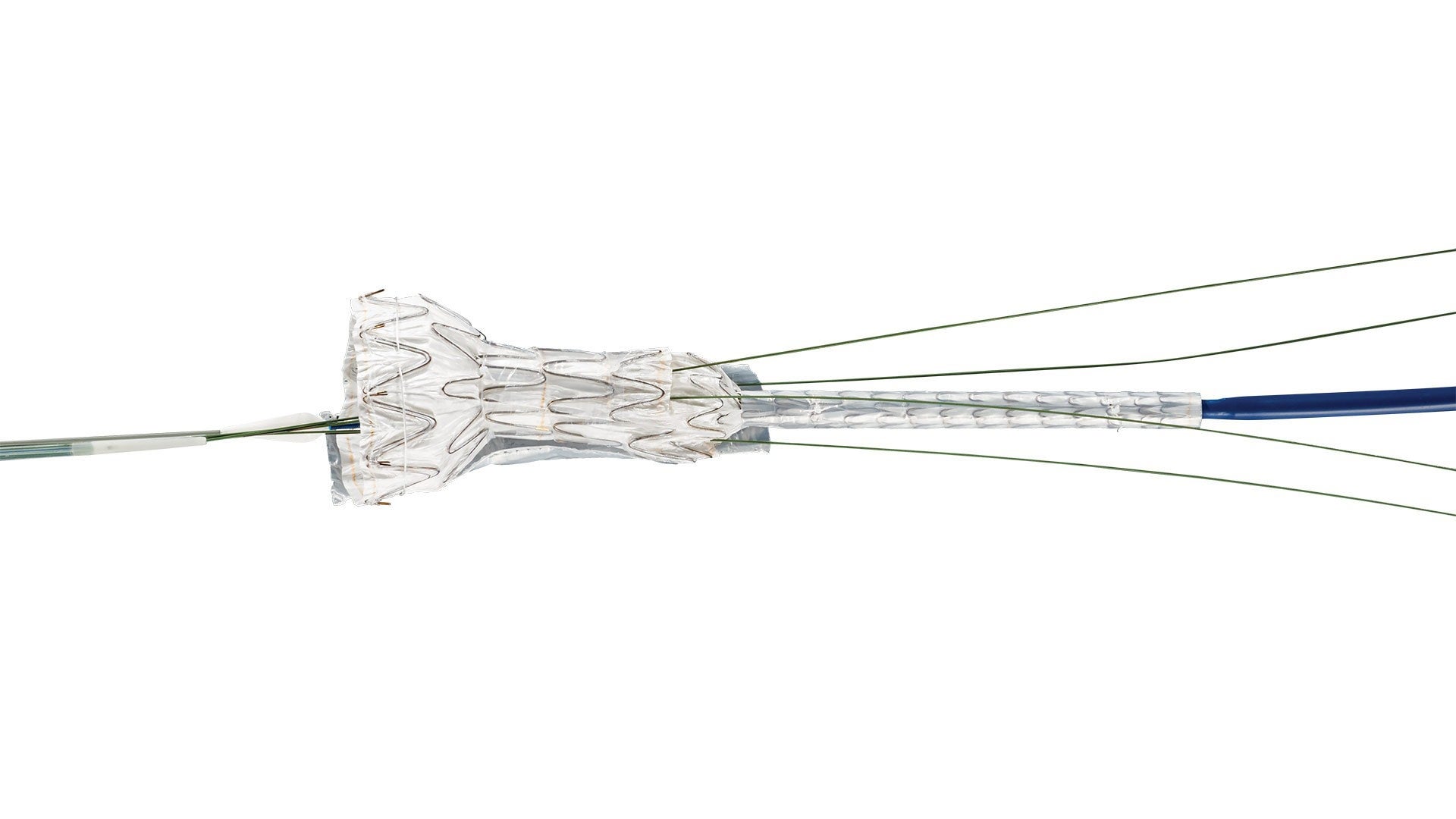
GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis (TAMBE)

Purpose built to manage complexity
At every step in the endovascular aortic repair process, TAMBE delivers. The complete system features rigorously tested components and the least aortic coverage among off-the-shelf BEVAR devices.1
Designed to maximize flexibility2
Deploy with confidence in complexity
The TAMBE Aortic Component offers a unique combination of deployment features designed to support greater perioperative flexibility when treating complex visceral aortic disease, including:
- Pre-cannulated portals to facilitate efficient branch vessel access
- Staged deployment to support space for cannulation and stepwise control
- The ability to reconstrain and reposition to optimize position prior to completing deployment
Gore offers dedicated support you can depend on in complex procedures
Training needs
Essential technical support with a deep reservoir of product knowledge
Case support
Field representatives from diverse clinical backgrounds support hundreds of aortic cases per year.
Purposeful innovation
We’re driven by something different. Because what’s at stake is everything.
Related to this product
- Bertoglio L, Grandi A, Carta N, et al. Comparison of anatomic feasibility of three different multibranched off-the-shelf stent-grafts designed for thoracoabdominal aortic aneurysms. J Vasc Surg. 2021;74(5):1472-1482.e4. doi:10.1016/j.jvs.2021.04.066.
- W. L. Gore & Associates. GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis Instructions for Use. W. L. Gore & Associates; 2023. MD193085.
- Farber, Mark A. et al. 1-year Results from the Pivotal Trial of a 4-Branch Thoracoabdominal Branch Endoprosthesis. Journal of Vascular Surgery, Volume 81, Issue 6, e236.

Refer to Instructions for Use at eifu.goremedical.com for a complete description of all applicable indications, warnings, precautions and contraindications for the markets where this product is available. RXOnly
INDICATIONS FOR USE IN EUROPE: The GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis is indicated for endovascular repair in patients with thoracoabdominal aortic aneurysms and pararenal aortic aneurysms who have appropriate anatomy.
CONTRAINDICATIONS: The GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis is contraindicated in patients with known sensitivities or allergies to TAMBE materials including ePTFE FEP nickel titanium alloy (Nitinol) stainless steel and gold. It is also contraindicated in patients who have a condition that threatens to infect the graft. Additionally patients with known hypersensitivity to heparin — including those who have previously experienced heparin-induced thrombocytopenia (HIT) type II and cannot receive the GORE® VIABAHN® VBX Balloon Expandable Endoprosthesis — should not receive this device.