Percutaneous transluminal angioplasty (PTA) has significant limitations when treating Arteriovenous (AV) access lesions1
- Patency rates diminish with each successive angioplasty procedure.1,2
- In the REVISE Clinical Study, patients undergoing PTA who had a previous intervention at the target lesion had lower primary patency at 6 months when compared with patients with no prior interventions at the target lesion (29.2% vs. 43.9%).1
- In the REVISE Clinical Study, patients undergoing PTA who had a previous intervention at the target lesion had lower primary patency at 6 months when compared with patients with no prior interventions at the target lesion (29.2% vs. 43.9%).1
- The 2019 KDOQI guidelines for vascular access specify a target goal of ≤ 3 interventions annually to maintain access.3
- In support of achieving this goal, KDOQI recommends stent grafts in preference to angioplasty alone for recurrent clinically significant stenotic AV access lesions.
Before
Thrombosed left arm AV graft. 80% stenosis just past venous anastomosis.
After
Post-placement of 7 mm x 5 cm GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface*,†

Image courtesy of Jason Burgess, M.D. Used with permission.
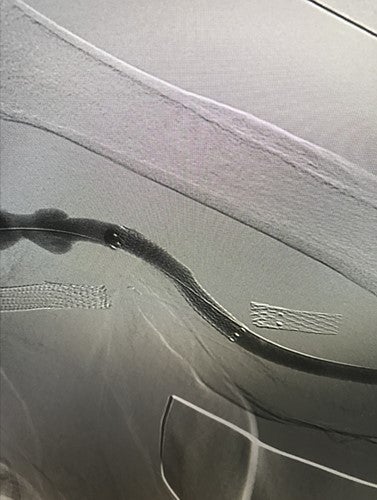
Image courtesy of Jason Burgess, M.D. Used with permission.
The VIABAHN® Device has proven patency independent of the number of prior interventions1
Proven patency
Provided consistent patency independent of the number of times a lesion has previously been treated.1
Fewer reinterventions
Reduced the average number of reinterventions by 27% over 2 years within the circuit compared to PTA.4
Stent graft design provides unique benefits to the treatment of lesions refractory to PTA
Expanded PTFE (ePTFE) flow lumen covers and seals off diseased tissue.

* As used by Gore, Heparin Bioactive Surface refers to Gore’s proprietary CBAS® Heparin Surface.
† Also referred to as the GORE® VIABAHN® Endoprosthesis with PROPATEN Bioactive Surface in some regions.
- Vesely T, DaVanzo W, Behrend T, Dwyer A, Aruny J. Balloon angioplasty versus Viabahn stent graft for treatment of failing or thrombosed prosthetic hemodialysis grafts. Journal of Vascular Surgery 2016;64(5):1400-1410.e1. http://www.sciencedirect.com/science/article/pii/S0741521416301756
- Kanterman RY, Vesely TM, Pilgram TK, Guy BW, Windus DW, Picus D. Dialysis access grafts: anatomic location of venous stenosis and results of angioplasty. Radiology 1995;195(1):135-139.
- Lok CE, Huber TS, Lee T, et al; KDOQI Vascular Access Guideline Work Group. KDOQI Clinical Practice Guideline for Vascular Access: 2019 update. American Journal of Kidney Diseases 2020;75(4)Supplement 2:S1-S164.
- Mohr BA, Sheen AL, Roy-Chaudhury P, Schultz SR, Aruny JE; REVISE Investigators. Clinical and economic benefits of stent grafts in dysfunctional and thrombosed hemodialysis access graft circuits in the REVISE Randomized Trial. Journal of Vascular & Interventional Radiology 2019;30(2):203-211.e4.

Refer to Instructions for Use at eifu.goremedical.com for a complete description of all applicable indications, warnings, precautions and contraindications for the markets where this product is available. RXOnly
INDICATIONS FOR USE IN THE U.S.: The GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface is indicated for improving blood flow in patients with symptomatic peripheral arterial disease in superficial femoral artery de novo and restenotic lesions up to 270 mm in length with reference vessel diameters ranging from 4.0 – 7.5 mm. The GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface is indicated for improving blood flow in patients with symptomatic peripheral arterial disease in superficial femoral artery in-stent restenotic lesions up to 270 mm in length with reference vessel diameters ranging from 4.0 – 6.5 mm. The GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface is indicated for improving blood flow in patients with symptomatic peripheral arterial disease in iliac artery lesions up to 80 mm in length with reference vessel diameters ranging from 4.0 – 12 mm. The GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface is also indicated for the treatment of stenosis or thrombotic occlusion at the venous anastomosis of synthetic arteriovenous (AV) access grafts.
CONTRAINDICATIONS: The GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface is contraindicated for non-compliant lesions where full expansion of an angioplasty balloon catheter was not achieved during pre-dilatation, or where lesions cannot be dilated sufficiently to allow passage of the delivery system. Do not use the GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface in patients with known hypersensitivity to heparin, including those patients who have had a previous incident of Heparin-Induced Thrombocytopenia (HIT) type II.