GORE® TAG® Thoracic Branch Endoprosthesis
A simplified solution within reach

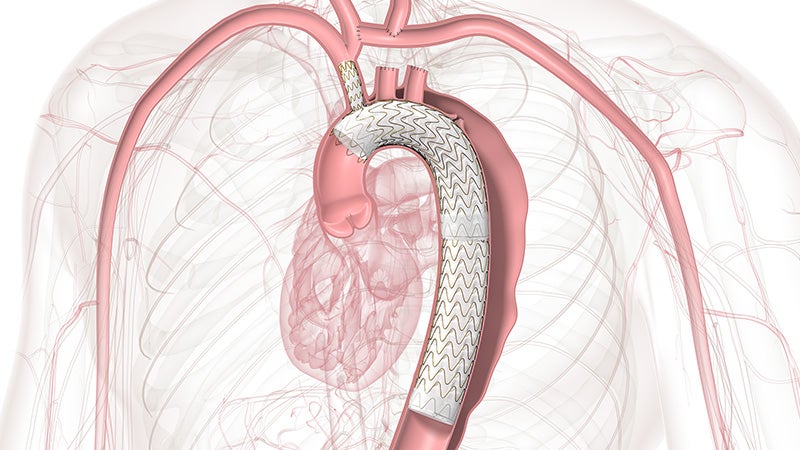
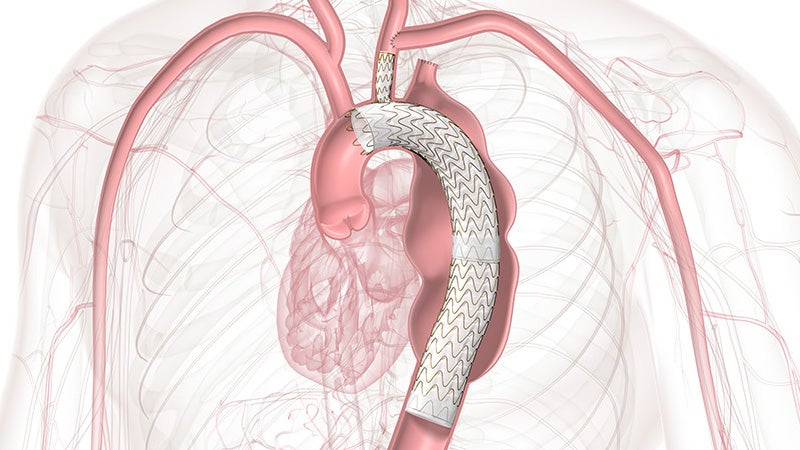
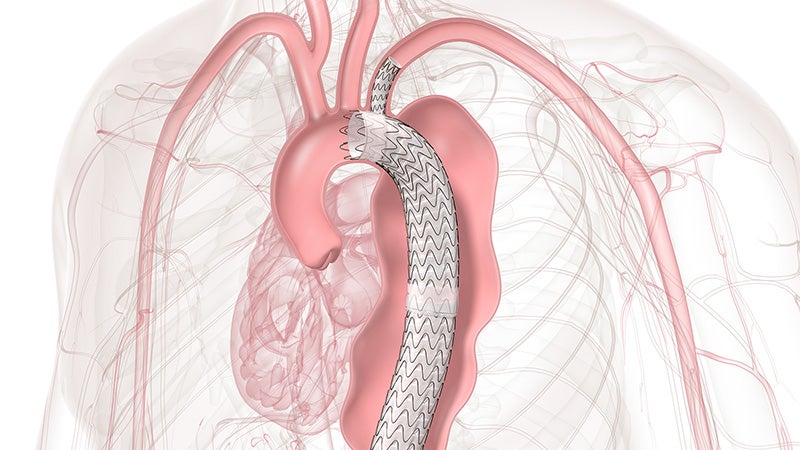
Approved across all aortic arch zones
The off-the-shelf, single branch device physicians trust for Zone 2 branched TEVAR procedures is now also approved for Zones 0 and 1, expanding minimally invasive repair of all lesions involving the aortic arch.
TBE provides an alternative to open surgical repair and reduces the overall impact of procedures like sternotomy, cardiopulmonary bypass and circulatory arrest.
Side branch patency through 1 year1
Zone 0: n = 0 occlusions
Zone 1: n = 1 occlusiona
Overall disabling stroke rate through 30 days1
TBE offers a demonstrated solution that preserves flow to the left subclavian artery (LSA) without the potential risks and complexity of surgical revascularization.
LSA branch patency through 1 year1
Disabling stroke rate, in aneurysm cohort, through 12 months1
Whether it’s Zone 0, 1 or 2, consider TBE for your next aortic arch repair.
How single-branch technology impacted one patient’s life.
Reimbursement & Coding Resources
On-demand, CEU-eligible coding webinars
Explore the latest changes to CPT and MS-DRG codes for thoracic and abdominal aortic endovascular procedures.
- TBE and TEVAR Physician Coding Webinar – Register with ZHealth to access recorded session.
- Hospital Coding Webinar – Register with Libman Education to access recorded session.
More reimbursement & coding resources
Coding is complex. Accuracy is critical. Stay precise with these tools:
- Access the Aortic Arch Repair Reimbursement Guide (PDF) for the latest updates in reimbursement and coding.
- Download our 2026 Hospital Inpatient Coding Guide (PDF) for in-depth coding details
Contact Us:
Gore Field Reimbursement Directors at + 1-800-248-8489 or fieldreimbursementdirectors@wlgore.com
or
REVENUE CYCLE CODING STRATEGIES® at + 1-888-812-0322
We're here to support you
Related to this product:
a Did not require intervention.
- GORE® TAG® Thoracic Branch Endoprosthesis [Instructions for Use] W. L. Gore & Associates; 2025. MD205141.

Refer to Instructions for Use at eifu.goremedical.com for a complete description of all applicable indications, warnings, precautions and contraindications for the markets where this product is available. RXOnly
INDICATIONS FOR USE: The GORE® TAG® Thoracic Branch Endoprosthesis is indicated for endovascular repair of lesions of the aortic arch and descending thoracic aorta, while maintaining flow into a single aortic arch branch vessel in patients who have: Adequate iliac/femoral access; Proximal Aortic Landing Zones: For Isolated Lesion Patients: Proximal landing zone cannot be aneurysmal, dissected, heavily calcified or heavily thrombosed; For Dissection Patients: Primary entry tear must be distal to the target branch vessel and the proximal extent of the landing zone must not be dissected; Aortic inner diameter range 16-42 mm; Proximal segment length (length from distal edge of target branch vessel to the midpoint of any proximal branch vessel) of at least 2.0-4.0 cm, depending on Aortic Component selection; Proximal covered length (measured from distal edge of target branch vessel to the distal edge of any proximal branch vessel) of at least 15–36 mm, depending on Aortic Component selection; For patients with prior ascending aorta or aortic arch repair with surgical graft: at least 2 cm landing zone proximal to the distal anastomosis; Target Branch Vessel Landing Zone: Landing zone cannot be aneurysmal, dissected, heavily thrombosed and severely tortuous (180 degree turn within the treated length); Target branch vessel inner diameter of 6–18 mm, depending on Side Branch Portal diameter selected; Target branch vessel minimum length of 2.5–3.0 cm, depending on Side Branch Portal diameter selected. Distal Landing Zone (Isolated Lesion Patients only): Outer curve length must be ≥ 2 cm proximal to celiac artery; Aortic inner diameter range 16-42 mm; Cannot be aneurysmal, dissected, heavily calcified or heavily thrombosed; Native Aorta or previously placed GORE® TAG® Conformable Thoracic Stent Graft.
CONTRAINDICATIONS: The GORE® TAG® Thoracic Branch Endoprosthesis is contraindicated in: Patients with known sensitivities or allergies to the device materials [ePTFE (polytetrafluoroethylene), FEP (fluoroethylpropylene), Nitinol (nickel, titanium), Gold, SB Component only - Heparin (CBAS® Heparin Surface)]; Patients who have a condition that threatens to infect the graft; Patients with known hypersensitivity to heparin, including those patients who have had a previous incident of Heparin-Induced Thrombocytopenia (HIT) type II.