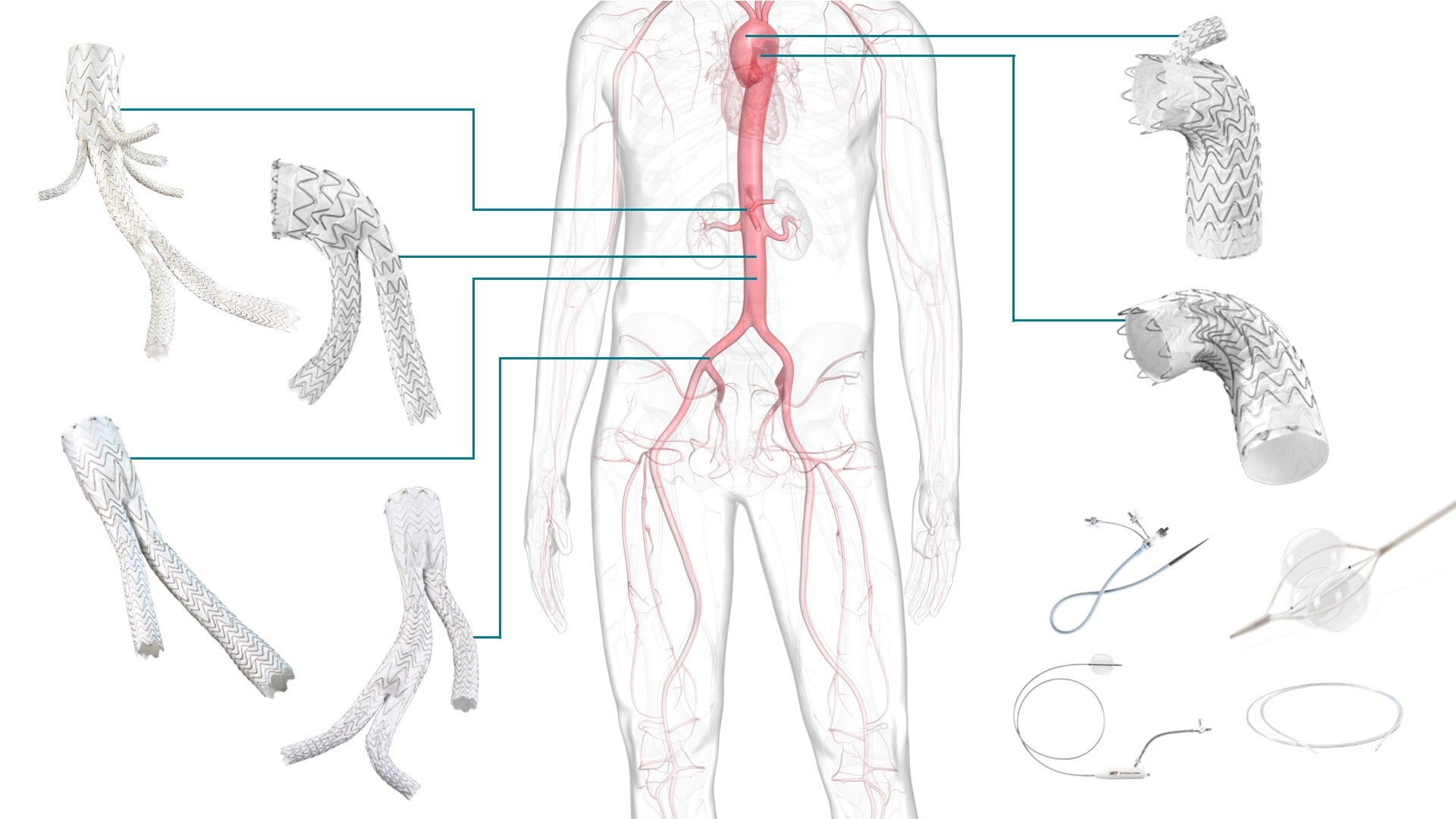
Enabling excellence in aortic care
Trusted expertise with comprehensive, high-quality aortic offerings and reliable support
Delivering cutting-edge aortic care with breakthrough endovascular solutions.
We’re dedicated to providing the most comprehensive aortic offering in the U.S. for patient’s needs.
Watch how Gore’s commitment to innovation and proven technology enabled a durable lifesaving repair to maintain an active lifestyle.
590,000
Patients treated worldwide with a AAA stent grafta
350,000
More than 350,000 TEVAR devices distributed worldwidea
55,000
Over 55,000 patients treated worldwidea with an iliac branch device
TWO
Global real-world evidence registries; GREAT and TOGETHER Registries
Legacy of innovation
2002
- FDA approval of the GORE® EXCLUDER® AAA Endoprosthesis
2003
- Nine new Contralateral Leg configurations added for GORE® EXCLUDER® AAA Endoprosthesis
2004
- Three new Trunk-Ipsilateral Leg configurations added for GORE® EXCLUDER® AAA Endoprosthesis
2005
- Six new Trunk-Ipsilateral Leg configurations added for GORE® EXCLUDER® AAA Endoprosthesis
2009
- FDA approval of new 31 mm Trunk-Ipsilateral Leg configurations for GORE® EXCLUDER® AAA Endoprosthesis
2010
- FDA approval of new EVAR delivery system allowing for controlled deployment and repositioning of Trunk-Ipsilateral Leg for GORE® EXCLUDER® AAA Endoprosthesis featuring C3® Delivery System
2011
- Introduction of new Contralateral Leg 23 mm and 27 mm diameters for GORE® EXCLUDER® AAA Endoprosthesis
2012
- Reduced profile available for larger Trunk-Ipsilateral Leg and Contralateral Leg configurations for GORE® EXCLUDER® AAA Endoprosthesis
- FDA approval of new 35 mm Trunk-Ipsilateral Leg configurations for GORE® EXCLUDER® AAA Endoprosthesis
2014
- New profile reduction for Trunk-Ipsilateral Leg configurations, Aortic Extenders for GORE® EXCLUDER® AAA Endoprosthesis
2020
- FDA approval of GORE® EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System, the only EVAR device to feature angulation control
2024
- First EVAR device approved to treat challenging neck anatomy 90° and 10 mm neck indication for the GORE® EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System
1997
- First thoracic device to enter clinical trials in U.S. for GORE® TAG® Thoracic Endoprosthesis
2005
- First FDA approved TEVAR device, GORE® TAG® Thoracic Endoprosthesis
2011
- FDA approval for Conformable GORE® TAG® Thoracic Endoprosthesis for treatment of aneurysms
2012
- FDA approval for the first thoracic device, Conformable GORE® TAG® Thoracic Endoprosthesis, for isolated lesions including traumatic transections
2013
- FDA approval for the first thoracic stent graft, Conformable GORE® TAG® Thoracic Endoprosthesis, for Type B dissections
2019
- FDA approval of first thoracic stent graft, GORE® TAG® Conformable Thoracic Stent Graft with ACTIVE CONTROL System, designed to provide staged deployment and angulation control
2024
- FDA approval of large diameter taper sizes (28 mm to 45 mm diameters)
2016
- First FDA-approved iliac branch device, GORE® EXCLUDER® Iliac Branch Endoprosthesis (IBE)
2022
- First flexible, off-the-shelf single-brand thoracic endoprosthesis, GORE® TAG® Thoracic Branch Endoprosthesis (TBE), for patients requiring Zone 2 treatment
2024
- First off-the-shelf device FDA approved to treat thoracoabdominal aortic aneurysms, GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis (TAMBE)
2025
- First flexible, off-the-shelf single-brand thoracic endoprosthesis for patients requiring Zone 0/1 treatment (TBE)
Quality aortic solutions built to endure
25+ years
Most studied EVAR deviceb (GORE® EXCLUDER® AAA Endoprosthesis) 25+ years of clinical experience
1,500+
Over 1,500 peer-reviewed publications
25+ years
25 years of clinical history
10
More than 10 clinical studies
Professional insights
Getting the most out of innovation: a Sg2 podcast
In this episode of Sg2 Perspectives, host Jayme Zage, PhD, is joined by Walter Holbein, Consulting Director at Sg2, and Matt Sondreaal, Abdominal Category Leader for W. L. Gore & Associates, to discuss how clinical innovation in abdominal aortic aneurysm (AAA) care can enhance health system performance. They explore the value of screening, the importance of training care teams and how minimally invasive procedures are transforming patient outcomes.
Seamless support, inside the operating room and out
From pre-case planning to reimbursement optimization, we collaborate so you can focus on outcomes.
Training Needs
Essential technical support with a deep reservoir of product knowledge.
Support
Our field representatives support hundreds of aortic cases per year, building on diverse clinical backgrounds.
Focused on Outcomes
Non-commissioned sales force - Our focus is on outcomes.
a Data on file 2025; W. L. Gore & Associates, Inc; Flagstaff, AZ.
b Based on company-sponsored trials and registries shown on clinicaltrials.gov for currently available stent grafts.
- Chaikof EL, Dalman RL, Eskandari MK, et al. The Society for Vascular Surgery practice guidelines on the care of patients with an abdominal aortic aneurysm. Journal of Vascular Surgery 2018;67(1):2‑77.e2.
- Moll FL, Powell JT, Fraedrich G, et al. European Society for Vascular Surgery. Management of abdominal aortic aneurysms clinical practice guidelines of the European Society for Vascular Surgery. European Journal of Vascular & Endovascular Surgery 2011;41(Supplement 1):S1‑S58.
- Schneider DB. One‑year U.S. results of GORER EXCLUDERR Iliac Branched Endograft: Advantages and limitations. Presented at the 42nd Annual Symposium on Vascular and Endovascular Issues, Techniques, Horizons, (VEITHsymposium); November 17‑21, 2015; New York, NY.

Refer to Instructions for Use at eifu.goremedical.com for a complete description of all applicable indications, warnings, precautions and contraindications for the markets where this product is available. RXOnly
GORE® EXCLUDER® AAA Endoprosthesis featuring C3® Delivery System
INDICATIONS FOR USE IN THE U.S.: Trunk-Ipsilateral Leg and Contralateral Leg Endoprosthesis. The GORE® EXCLUDER® AAA Endoprosthesis is intended to exclude the aneurysm from the blood circulation in patients diagnosed with infrarenal abdominal aortic aneurysm (AAA) disease and who have appropriate anatomy as described below: Adequate iliac/femoral access; Infrarenal aortic neck treatment diameter range of 19–32 mm and a minimum aortic neck length of 15 mm; Proximal aortic neck angulation ≤ 60°; Iliac artery treatment diameter range of 8–25 mm and iliac distal vessel seal zone length of at least 10 mm. Aortic Extender and Iliac Extender Endoprosthesis. The Aortic and Iliac Extender Endoprostheses are intended to be used after deployment of the GORE® EXCLUDER® AAA Endoprosthesis. These extensions are intended to be used when additional length and/or sealing for aneurysmal exclusion is desired.
CONTRAINDICATIONS: The GORE® EXCLUDER® AAA Endoprosthesis is contraindicated in: patients with known sensitivities or allergies to the device materials; patients with a systemic infection who may be at increased risk of endovascular graft infection.
GORE® EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System
INDICATIONS FOR USE IN THE U.S.: The GORE® EXCLUDER® Conformable AAA Endoprosthesis is intended to exclude the aneurysm from the blood circulation in patients diagnosed with infrarenal abdominal aortic aneurysm (AAA) disease and who have appropriate anatomy as described below: Adequate iliac/femoral access; Infrarenal aortic neck treatment diameter range of 16-32 mm and a minimum aortic neck length of 10 mm; Proximal aortic neck angulation ≤ 90°; Iliac artery treatment diameter range of 8-25 mm and iliac distal vessel seal zone length of at least 10 mm. Aortic Extender Endoprosthesis and Iliac Extender Endoprosthesis Components are intended to be used after deployment of the GORE® EXCLUDER® Conformable AAA Endoprosthesis. These extensions are intended to be used when additional length and/or sealing for aneurysmal exclusion is desired.
CONTRAINDICATIONS: The GORE® EXCLUDER® Conformable AAA Endoprosthesis is contraindicated in: patients with known sensitivities or allergies to the device materials. All components of the GORE® EXCLUDER® Conformable Endoprosthesis contain expanded polytetrafluoroethylene (ePTFE), fluorinated ethylene propylene (FEP), nitinol (nickel-titanium alloy) and gold. Patients with systemic infection who may be at increased risk of endovascular graft infection.
GORE® EXCLUDER® Iliac Branch Endoprosthesis (IBE)
INDICATIONS FOR USE IN THE U.S.: Iliac Branch and Internal Iliac Components. The GORE® EXCLUDER® Iliac Branch Endoprosthesis (IBE) is intended to be used with the GORE® EXCLUDER® AAA Endoprosthesis or the GORE® EXCLUDER® Conformable AAA Endoprosthesis to isolate the common iliac artery from systemic blood flow and preserve blood flow in the external iliac and internal iliac arteries in patients with a common iliac or aortoiliac aneurysm, who have appropriate anatomy, including: adequate iliac/ femoral access; minimum common iliac diameter of 17 mm at the proximal implantation zone of the IBE; external Iliac artery treatment diameter range of 6.5–25 mm and seal zone length of at least 10 mm; internal iliac artery treatment diameter range of 6.5–13.5 mm and seal zone length of at least 10 mm; adequate length from the lowest major renal artery to the internal iliac artery to accommodate the total endoprosthesis length, calculated by adding the minimum lengths of required components, taking into account appropriate overlaps between components. GORE® EXCLUDER® AAA Endoprosthesis Components used in conjunction with GORE® EXCLUDER® Iliac Branch Endoprosthesis. Trunk-Ipsilateral Leg Component. The Trunk-Ipsilateral Leg is intended to provide proximal seal and fixation for the endovascular repair of the aneurysm. For more information on the Trunk-Ipsilateral Leg Component indications for use and deployment, see the GORE® EXCLUDER® AAA Endoprosthesis or the GORE® EXCLUDER® Conformable Endoprosthesis Instructions for Use. Contralateral Leg Endoprosthesis Component. The Contralateral Leg Endoprosthesis is intended to bridge the GORE® EXCLUDER® Device Trunk-Ipsilateral Component to the GORE® EXCLUDER® Iliac Branch Endoprosthesis following deployment of the GORE® EXCLUDER® Iliac Branch Endoprosthesis. Additionally, the Contralateral Leg Endoprosthesis is intended to be used for distal extension of the Iliac Branch Component in the external iliac artery. The Iliac Branch Component can treat external iliac artery diameters up to 13.5 mm. This ability to extend the Iliac Branch Component distally with any Contralateral Leg Endoprosthesis expands the external iliac artery treatment range up to 25 mm. For more information on the Trunk-Ipsilateral Leg and Contralateral Leg Endoprosthesis Component indications for use and deployment, see the GORE® EXCLUDER® AAA Endoprosthesis Instructions for Use. Aortic Extender and Iliac Extender Components. The Aortic and Iliac Extender Components can be used after deployment of the GORE® EXCLUDER® Iliac Branch and GORE® EXCLUDER® AAA Endoprostheses. These extensions are used when additional length and/or sealing for aneurysmal exclusion is desired. For more information on Aortic Extender and Iliac Extender indications for use and deployment, see the GORE® EXCLUDER® AAA Endoprosthesis or the GORE® EXCLUDER® Conformable Endoprosthesis Instructions For Use.
CONTRAINDICATIONS: The GORE® EXCLUDER® Iliac Branch Endoprosthesis is contraindicated in: patients with known sensitivities or allergies to the device materials. All components of the GORE® EXCLUDER® Iliac Branch Endoprosthesis and the GORE® EXCLUDER® AAA Endoprosthesis contain ePTFE, FEP, nitinol (nickel-titanium alloy) and gold; patients with a systemic infection who may be at increased risk of endovascular graft infection.
GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis (TAMBE)
INDICATIONS FOR USE IN THE U.S.: The GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis is indicated for endovascular repair in patients with thoracoabdominal aortic aneurysms and high-surgical risk patients with pararenal aortic aneurysms who have appropriate anatomy as described below: Adequate iliac/femoral access and brachial/axillary access; proximal (supraceliac) aortic neck treatment diameter range over 2 cm seal zone of 22-34 mm for aneurysms extending up to 6.5 cm or less above the origin of the most proximal branch vessel; aortic neck angle ≤ 60° at the Aortic Component proximal seal zone; iliac artery treatment diameter range of 8-25 mm and iliac artery seal zone length of at least 10 mm; renal artery seal zone diameters between 4.0-10.0 mm; celiac and superior mesenteric artery seal zone diameters between 5.0-12.0 mm; ≥ 15 mm seal zone length in renal arteries, superior mesenteric artery and celiac artery; and visceral segment of aorta (3 cm proximal through 9.5 cm distal to the most proximal visceral artery) must be ≥ 20 mm in diameter.
CONTRAINDICATIONS: The GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis is contraindicated in: Patients with known sensitivities or allergies to the TAMBE materials including ePTFE, FEP, nickel titanium alloy (Nitinol), stainless steel and gold. Patients who have a condition that threatens to infect the graft. Patients with known hypersensitivity to heparin, including patients who have had a previous incident of Heparin-Induced Thrombocytopenia (HIT) type II and cannot receive the GORE® VIABAHN® VBX Balloon Expandable Endoprosthesis.
GORE® TAG® Conformable Thoracic Stent Graft with ACTIVE CONTROL System
INDICATIONS FOR USE IN THE U.S.: The GORE® TAG® Conformable Thoracic Stent Graft is intended for endovascular repair of all lesions of the descending thoracic aorta, including: isolated lesions in patients who have appropriate anatomy, including: adequate iliac/femoral access, aortic inner diameter in the range of 16-42 mm, ≥ 20 mm non-aneurysmal aorta proximal and distal to the lesion; Type B dissections in patients who have appropriate anatomy, including: adequate iliac/femoral access, ≥ 20 mm landing zone proximal to the primary entry tear; proximal extent of the landing zone must not be dissected, diameter at proximal extent of proximal landing zone in the range of 16-42 mm.
CONTRAINDICATIONS: The GORE® TAG® Conformable Thoracic Stent Graft is contraindicated in: Patients with known sensitivities or allergies to the device materials; patients who have a condition that threatens to infect the graft.
GORE® TAG® Thoracic Branch Endoprosthesis (TBE)
INDICATIONS FOR USE IN THE U.S.: The GORE® TAG® Thoracic Branch Endoprosthesis is indicated for endovascular repair of lesions of the aortic arch and descending thoracic aorta, while maintaining flow into a single aortic arch branch vessel in patients who have: Adequate iliac/femoral access; Proximal Aortic Landing Zones: For Isolated Lesion Patients: Proximal landing zone cannot be aneurysmal, dissected, heavily calcified or heavily thrombosed; For Dissection Patients: Primary entry tear must be distal to the target branch vessel and the proximal extent of the landing zone must not be dissected; Aortic inner diameter range 16-42 mm; Proximal segment length (length from distal edge of target branch vessel to the midpoint of any proximal branch vessel) of at least 2.0-4.0 cm, depending on Aortic Component selection; Proximal covered length (measured from distal edge of target branch vessel to the distal edge of any proximal branch vessel) of at least 15–36 mm, depending on Aortic Component selection; For patients with prior ascending aorta or aortic arch repair with surgical graft: at least 2 cm landing zone proximal to the distal anastomosis; Target Branch Vessel Landing Zone: Landing zone cannot be aneurysmal, dissected, heavily thrombosed and severely tortuous (180 degree turn within the treated length); Target branch vessel inner diameter of 6–18 mm, depending on Side Branch Portal diameter selected; Target branch vessel minimum length of 2.5–3.0 cm, depending on Side Branch Portal diameter selected. Distal Landing Zone (Isolated Lesion Patients only): Outer curve length must be ≥ 2 cm proximal to celiac artery; Aortic inner diameter range 16-42 mm; Cannot be aneurysmal, dissected, heavily calcified or heavily thrombosed; Native Aorta or previously placed GORE® TAG® Conformable Thoracic Stent Graft.
CONTRAINDICATIONS: The GORE® TAG® Thoracic Branch Endoprosthesis is contraindicated in: Patients with known sensitivities or allergies to the device materials [ePTFE (polytetrafluoroethylene), FEP (fluoroethylpropylene), Nitinol (nickel, titanium), Gold, SB Component only - Heparin (CBAS® Heparin Surface)]; Patients who have a condition that threatens to infect the graft; Patients with known hypersensitivity to heparin, including those patients who have had a previous incident of Heparin-Induced Thrombocytopenia (HIT) type II.
GORE® DRYSEAL Flex Introducer Sheath
INDICATIONS FOR USE IN THE U.S.: The GORE® DRYSEAL Flex Introducer Sheath is intended to be inserted in the vasculature to provide a conduit for the insertion of endovascular devices while minimizing blood loss associated with such insertions.
CONTRAINDICATIONS: No known contraindications.
GORE® Molding & Occlusion Balloon Catheter
INDICATIONS FOR USE IN THE U.S.: The GORE® Molding and Occlusion Balloon Catheter is intended for temporary occlusion of large diameter vessels or to assist the expansion of self-expanding endovascular prostheses (stent grafts).
CONTRAINDICATIONS: The GORE® Molding and Occlusion Balloon Catheter is contraindicated in patients who: are contraindicated to contrast media or anticoagulants; have an arterial entry site that cannot accommodate a 10 Fr introducer sheath; are minors; are pregnant.
GORE® Tri-Lobe Balloon Catheter
INDICATIONS FOR USE IN THE U.S.: The GORE® Tri-Lobe Balloon Catheter is indicated to facilitate in the endovascular repair of the thoracic or abdominal aorta due to lesions including aneurysms, dissections, trauma, and penetrating aortic ulcers.
CONTRAINDICATIONS: There are no known contraindications.
GORE® Tri-Lumen Catheter
INDICATIONS FOR USE IN THE U.S.: The GORE® Tri-Lumen Catheter is a multi-lumen catheter indicated for use in endovascular procedures requiring multiple guidewires and through-and-through access, in which the catheter leading tip exits the patient, for the implantation of branched stent grafts. Standard techniques for placement of vascular access sheaths, catheters and wires should be employed.
CONTRAINDICATIONS: No known contraindications.